Vitamin D3 (Cholecalciferol): Biochemical Profile & Clinical Nutrition
Biochemical Profile
Classification: Fat-soluble Secosteroid (Prohormone); Subclass: Calciferols.
Chemical Structure/Properties: Cholecalciferol (Vitamin D3) is characterized by a broken steroid ring structure (seco-steroid). It is chemically distinct from Ergocalciferol (Vitamin D2) in its side-chain structure. Unlike classic vitamins acting as co-factors, Vitamin D3 functions primarily as a prohormone precursor. It is hydrophobic and requires lipid binding proteins (Vitamin D-binding protein, DBP) for systemic transport in the circulatory system.
Metabolic Function & Mechanism
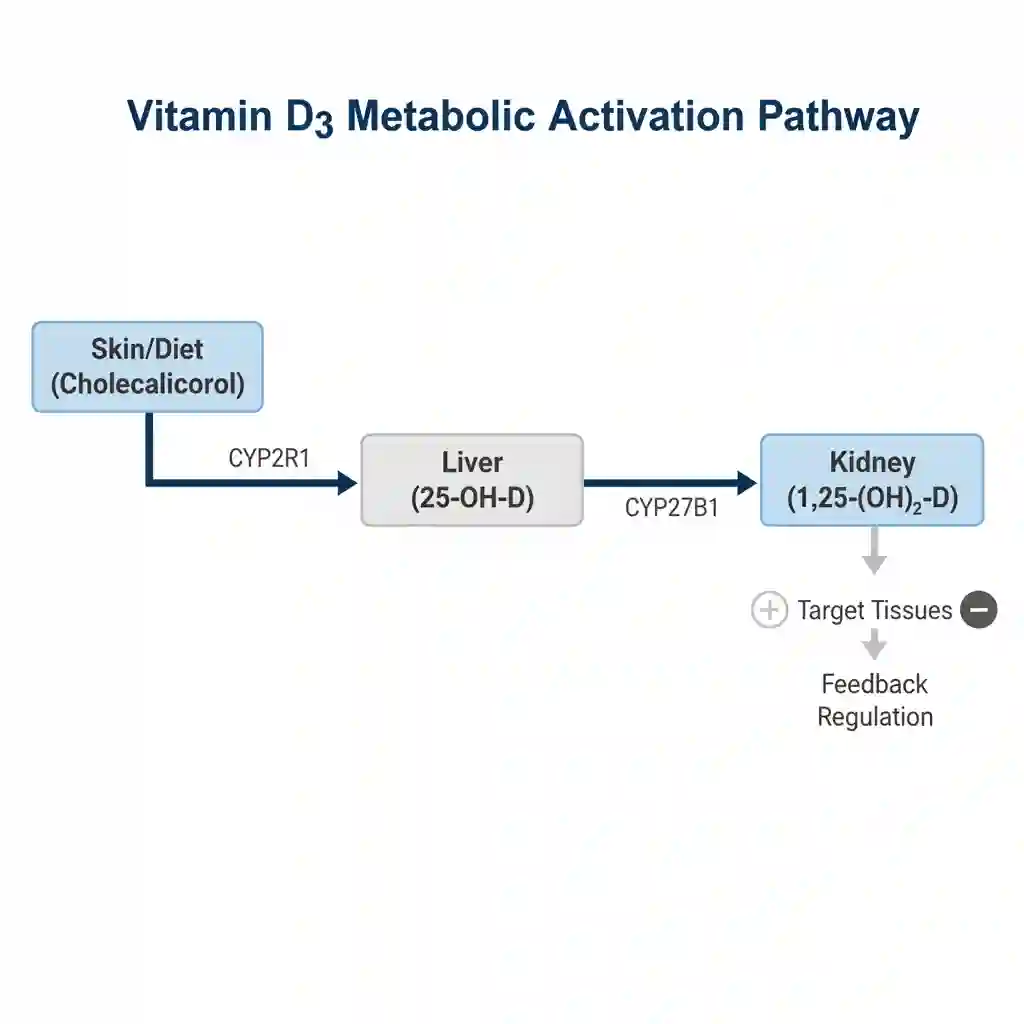
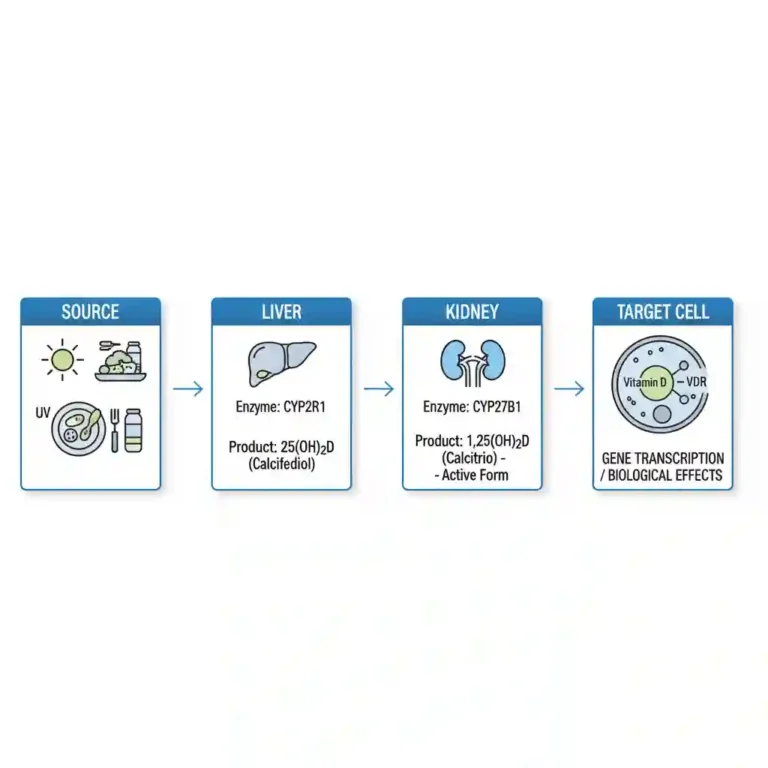
The metabolic activation of Vitamin D3 is a multi-step enzymatic process involving hydroxylation in the liver and kidneys:
- Absorption & Transport: Dietary Vitamin D3 is absorbed in the small intestine via passive diffusion and incorporated into chylomicrons, requiring dietary lipids and bile salts.
- Hepatic Hydroxylation (25-Hydroxylation): In the liver, the enzyme CYP2R1 (25-hydroxylase) converts Cholecalciferol into Calcifediol (25-hydroxyvitamin D [25(OH)D]). This is the major circulating form used for clinical status assessment.
- Renal Hydroxylation (1-alpha-Hydroxylation): In the proximal tubule of the kidney, the enzyme CYP27B1 converts 25(OH)D into the biologically active hormone Calcitriol (1,25-dihydroxyvitamin D [1,25(OH)2D]).
Cellular Mechanism: Calcitriol functions as a steroid hormone by binding to the nuclear Vitamin D Receptor (VDR). The VDR-Calcitriol complex heterodimerizes with the Retinoid X Receptor (RXR), binding to Vitamin D Response Elements (VDREs) in DNA to regulate the transcription of genes involved in calcium absorption (e.g., TRPV6, Calbindin).
Medical Nutrition Therapy (MNT) Applications
Clinical guidelines indicate specific therapeutic protocols for Vitamin D3 in the following pathophysiological states:
- Rickets & Osteomalacia: Indicated for correcting defective bone mineralization and restoring calcium homeostasis.
- Osteoporosis Management: Used as an adjunct therapy with Calcium and Bisphosphonates to suppress secondary hyperparathyroidism and reduce fracture risk.
- Chronic Kidney Disease (CKD – Stages 3-5): Due to impaired renal 1-alpha-hydroxylation, patients may require active Vitamin D analogs (Calcitriol) or high-dose Cholecalciferol to manage CKD-Mineral and Bone Disorder (CKD-MBD).
- Malabsorption Syndromes: Patients with Cystic Fibrosis, Crohn’s Disease, or Post-Bariatric Surgery require high-dose therapeutic supplementation due to impaired lipid absorption mechanisms.
Dietary Sources & Bioavailability
Natural dietary sources of Vitamin D3 are limited. Bioavailability is strictly dependent on the presence of concomitant dietary fat.
| Source Category | Primary Examples | Bioavailability & Absorption Efficiency |
|---|---|---|
| Marine Sources | Cod liver oil, Swordfish, Salmon, Mackerel | High. Naturally occurs in a lipid matrix, facilitating micelle formation and chylomicron transport. |
| Fortified Foods | Cow’s milk, Plant-based milk alternatives, Orange juice | Variable. Bioavailability depends on the fat content of the carrier vehicle (e.g., skim milk vs. whole milk). |
| Supplements | Softgels (Oil-based) vs. Tablets (Powder) | High (Oil-based). Studies suggest oil-based softgels offer superior absorption compared to dry tablets taken without a fatty meal. |
| Endogenous Synthesis | UVB Radiation (290–315 nm) on Skin | Variable. Dependent on melanin content, latitude, and age. Bypasses intestinal absorption issues but regulated by thermal isomerization to prevent toxicity. |
Safety & Interactions
While Vitamin D3 has a wide therapeutic window, supraphysiological doses can lead to toxicity.
- Contraindications: Pre-existing Hypercalcemia and Hypervitaminosis D.
- Drug-Nutrient Interactions:
- Corticosteroids: Can reduce calcium absorption and impair Vitamin D metabolism.
- Orlistat/Cholestyramine: Lipid-lowering drugs and lipase inhibitors significantly reduce the absorption of fat-soluble vitamins, including D3.
- Thiazide Diuretics: May decrease urinary calcium excretion; concurrent high-dose Vitamin D increases risk of hypercalcemia.
- Toxicity Profile: Chronic excess leads to hypercalcemia, resulting in soft tissue calcification (calcinosis) and renal failure.