Clinical Definition
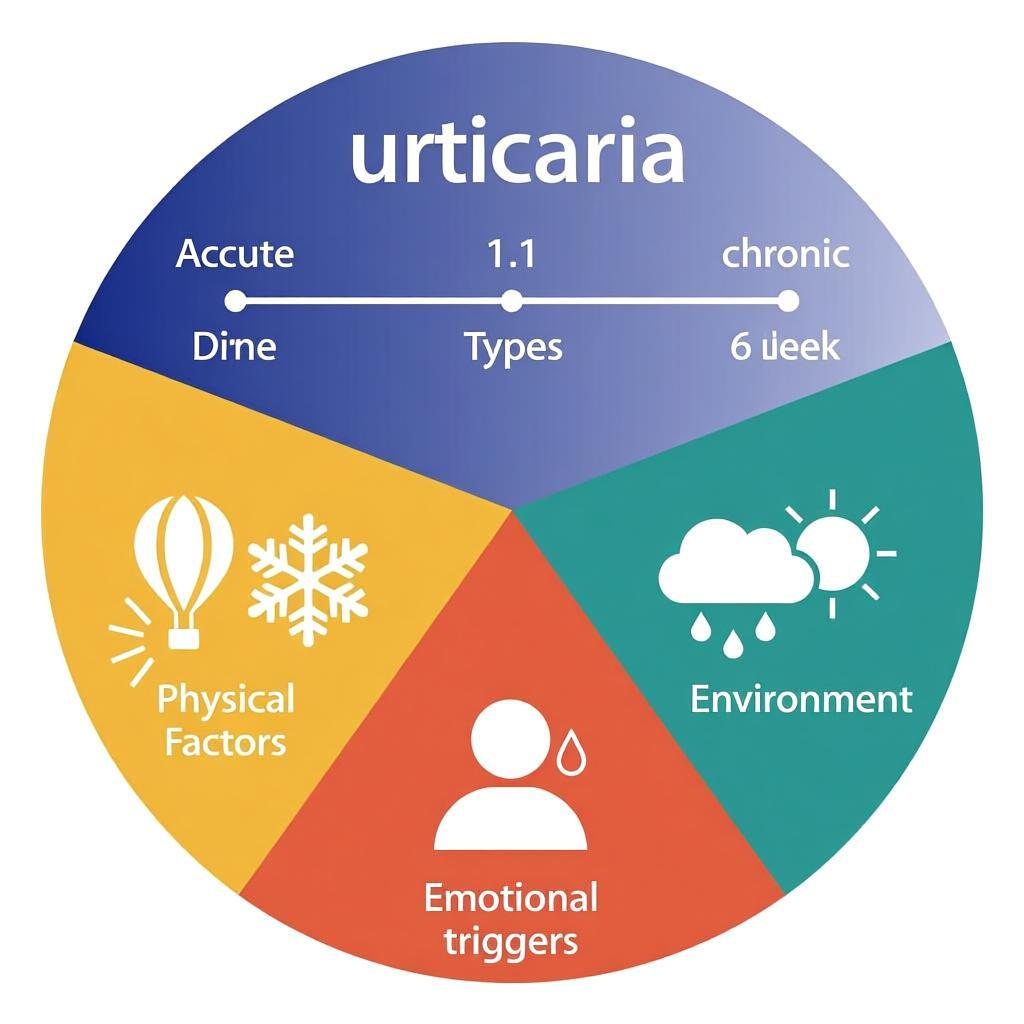
Urticaria is a mast cell-driven vascular reaction of the skin characterized by the development of wheals (hives), angioedema, or both. A wheal is defined as a superficial, pruritic, evanescent swelling of the skin that typically resolves within 24 hours without scarring. Clinically, it is classified based on duration into Acute Urticaria (< 6 weeks) and Chronic Urticaria (> 6 weeks), with the latter further divided into Spontaneous (CSU) and Inducible (CIndU) subtypes.
Clinical Coding & Classification
| System / Category | Code(s) | Description |
|---|---|---|
| ICD-10-CM | L50.1 | Idiopathic urticaria (Chronic Spontaneous Urticaria – CSU) |
| ICD-10-CM | L50.0 | Allergic urticaria |
| ICD-10-CM | T78.3 | Angioneurotic edema (Angioedema) |
| CPT (Therapeutic) | 96372 | Therapeutic, prophylactic, or diagnostic injection (subcutaneous or intramuscular); often used for biologic administration |
| CPT (Lab) | 86003 | Allergen specific IgE; quantitative or semiquantitative |
Epidemiology & Statistics
The lifetime prevalence of any form of urticaria is approximately 20%. Chronic Spontaneous Urticaria (CSU) affects 0.5% to 1% of the global population. While acute urticaria is common in children, chronic urticaria is more prevalent in adults, particularly women aged 20 to 40 years (female-to-male ratio of 2:1). Angioedema accompanies wheals in approximately 40% of CSU cases.
Pathophysiology (Mechanism)
The central effector cell is the cutaneous mast cell. Activation leads to degranulation and release of inflammatory mediators:
1. Histamine Release: Stimulates H1 receptors on endothelial cells, causing vasodilation (erythema) and plasma leakage (edema/wheal), and sensory nerve stimulation (pruritus).
2. Autoimmunity (Type IIb): In 30-40% of CSU patients, IgG autoantibodies are directed against the high-affinity IgE receptor (FcεRIα) or against IgE itself, leading to chronic mast cell activation.
3. Bradykinin: Primarily involved in hereditary or ACE-inhibitor-induced angioedema, distinct from histamine-mediated pathways.
Standard Management Protocols
Therapy follows the EAACI/WAO/GA²LEN international guidelines, characterized by a stepwise approach.
- Pharmacological Classes:
- Second-Generation H1-Antihistamines: (e.g., Bilastine, Desloratadine) First-line therapy. Guidelines recommend up-dosing to 4-fold the standard dose for non-responders.
- Biologics (Anti-IgE): (e.g., Omalizumab) A recombinant DNA-derived humanized monoclonal antibody that binds to free IgE. Indicated for CSU refractory to antihistamines.
- Immunosuppressants: (e.g., Cyclosporine A) Third-line option for recalcitrant cases, inhibiting calcineurin-phosphatase.
- Diagnostic Evaluation:
- Urticaria Activity Score (UAS7): Standardized tool for assessing disease activity and therapeutic response.
Healthcare Resource Utilization
CSU imposes a significant socioeconomic burden:
- Biologic Costs: High annual cost of monoclonal antibody therapy for refractory patients.
- Emergency Utilization: Frequent ER presentations due to fear of anaphylaxis, particularly when angioedema involves the oropharynx.