Clinical Definition
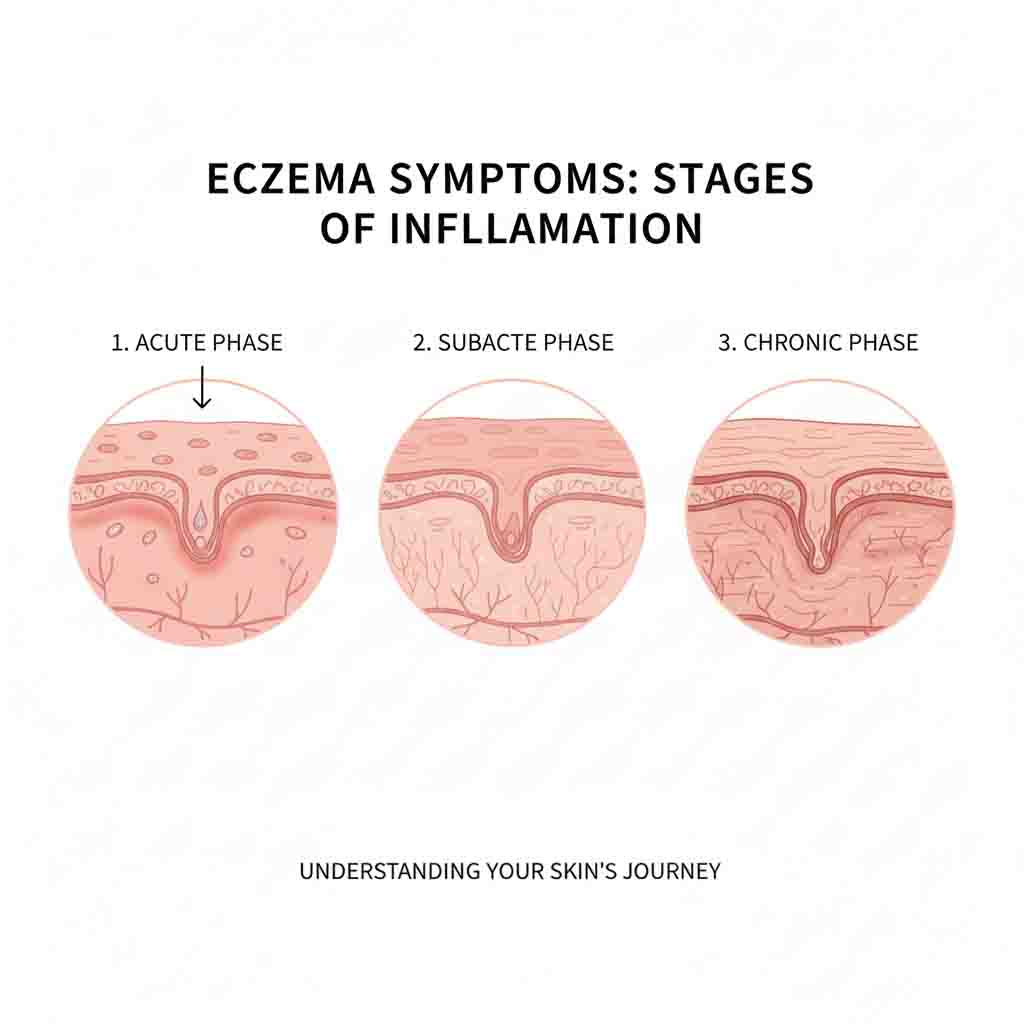
Eczema, clinically referred to as Atopic Dermatitis (AD), is a chronic, relapsing, pruritic inflammatory dermatosis. It involves a complex interplay of skin barrier dysfunction (e.g., Filaggrin mutations) and immune dysregulation (Th2 pathway activation). The condition manifests morphologically in stages: acute (vesiculation, erythema), subacute (excoriation, crusting), and chronic (lichenification). It is the cutaneous manifestation of the “Atopic March,” often associated with asthma and allergic rhinitis.
Clinical Coding & Classification
| System / Category | Code(s) | Description |
|---|---|---|
| ICD-10-CM | L20.9 | Atopic dermatitis, unspecified |
| ICD-10-CM | L23.9 | Allergic contact dermatitis, unspecified cause |
| CPT (Therapeutic) | 96910 | Photochemotherapy; tar and ultraviolet B (Goeckerman treatment) or petrolatum and ultraviolet B |
| CPT (Diagnostic) | 86003 | Allergen specific IgE; quantitative or semiquantitative, each allergen |
| Affected System | Integumentary; Immunological | Epidermal Barrier; Th2 Immune Pathway |
Epidemiology & Statistics
Atopic Dermatitis affects approximately 15-20% of children and 1-3% of adults globally. The prevalence has increased 2- to 3-fold in industrialized nations over the past three decades. Onset typically occurs in early childhood, with 60% of cases presenting within the first year of life. Adult-onset AD is recognized as a distinct phenotype often requiring systemic therapy.
Pathophysiology (Mechanism)
The pathogenesis is multifactorial, centering on the “Outside-Inside” vs. “Inside-Outside” hypotheses:
1. Epidermal Barrier Defect: Null mutations in the Filaggrin (FLG) gene lead to a deficiency in natural moisturizing factors, increasing transepidermal water loss (TEWL) and allowing allergen/pathogen penetration.
2. Immune Dysregulation: Penetration of allergens triggers a type 2 helper T-cell (Th2) response, releasing proinflammatory cytokines IL-4 and IL-13. This cascade suppresses antimicrobial peptides and drives inflammation.
3. Pruritus-Scratch Cycle: Inflammation sensitizes cutaneous nerve fibers, leading to scratching, which further damages the barrier and perpetuates inflammation.
Standard Management Protocols
Therapy follows a stepwise approach based on severity (e.g., EASI Score).
- Topical Therapies:
- Topical Corticosteroids (TCS): The mainstay for acute flares (varying potencies).
- Topical Calcineurin Inhibitors (TCI): (e.g., Tacrolimus, Pimecrolimus) Non-steroidal agents for sensitive areas (face, folds).
- PDE4 Inhibitors: (e.g., Crisaborole) For mild-to-moderate disease.
- Systemic/Biologic Therapies (Moderate-to-Severe):
- Biologics: (e.g., Dupilumab) Monoclonal antibodies targeting the IL-4/IL-13 receptor.
- JAK Inhibitors: (e.g., Upadacitinib, Abrocitinib) Oral small molecules blocking the Janus Kinase pathway to inhibit cytokine signaling.
Healthcare Resource Utilization
AD imposes a substantial burden due to:
- Direct Costs: High annual cost of novel biologic therapies and frequent dermatology visits.
- Quality of Life: Severe pruritus causes sleep disturbance and psychosocial morbidity, impacting workforce productivity.