Metformin Hydrochloride: Pharmacological Profile & Clinical Data
Pharmacological Classification
Class: Biguanide Antihyperglycemic Agent.
ATC Code: A10BA02 (Alimentary tract and metabolism > Drugs used in diabetes > Blood glucose lowering drugs, excluding insulins > Biguanides).
Common Trade Names: Glucophage, Glumetza, Fortamet (Reference only; availability varies by region).
Mechanism of Action (MOA)
Metformin acts primarily as an insulin sensitizer that alters cellular energy metabolism. Unlike sulfonylureas, it does not stimulate insulin secretion (non-secretagogue) and therefore carries a minimal risk of hypoglycemia when used as monotherapy. The molecular mechanism involves multiple pathways:
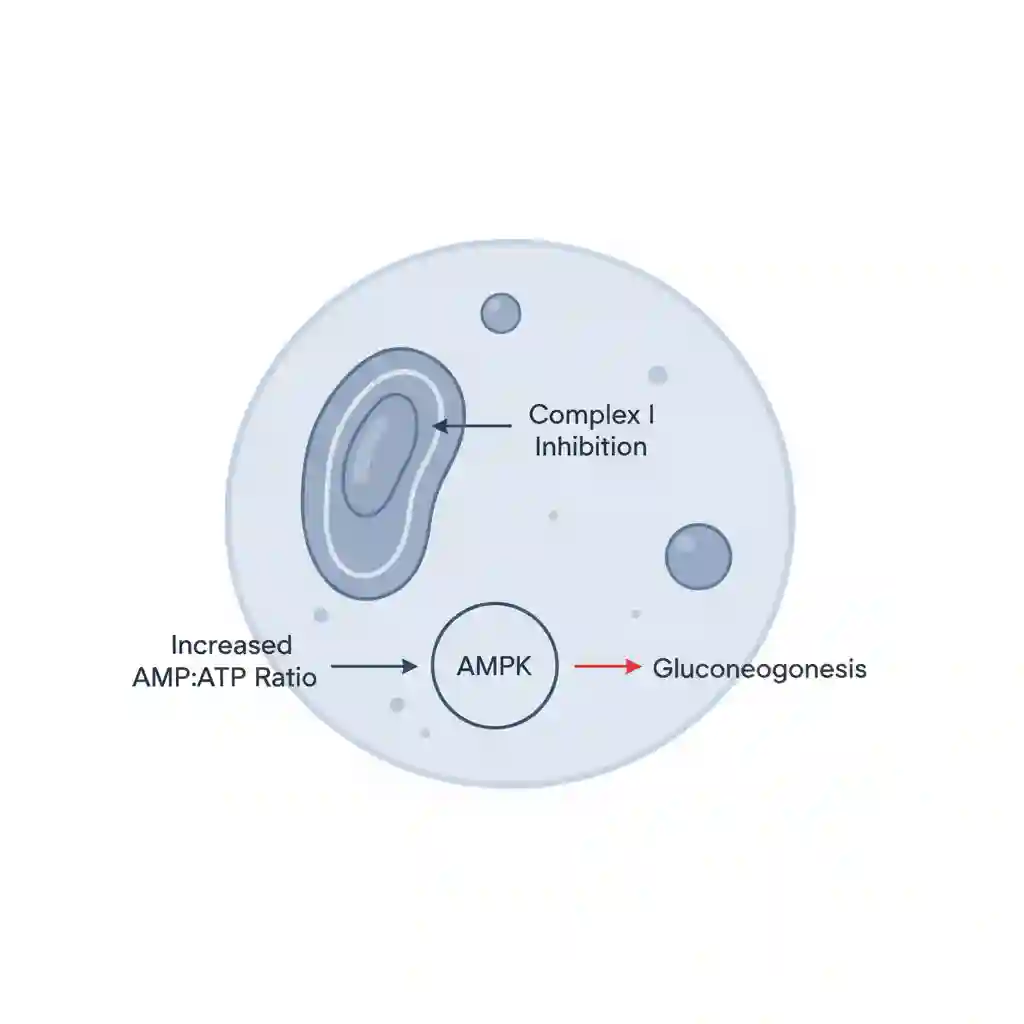
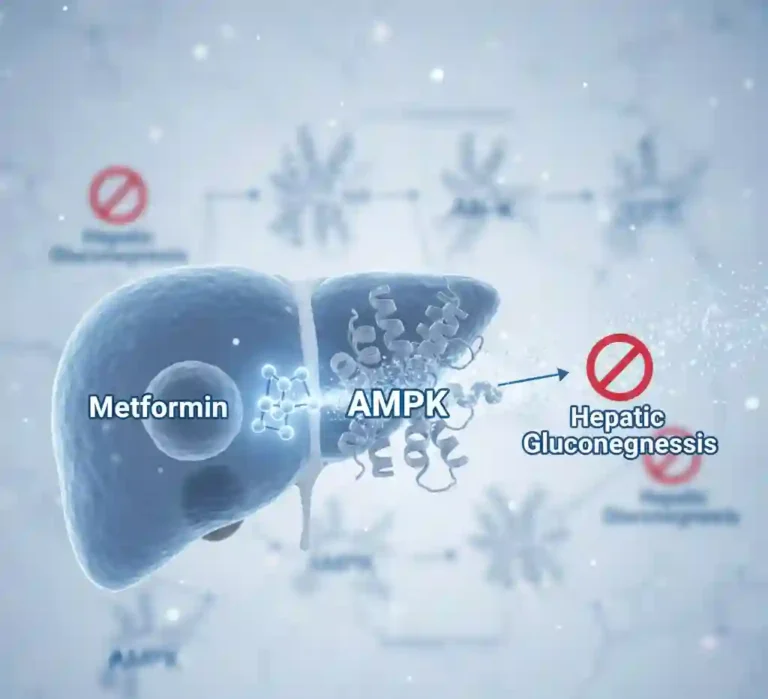
- Inhibition of Hepatic Gluconeogenesis: Metformin inhibits the mitochondrial respiratory chain (Complex I), leading to an increased AMP/ATP ratio. This activates AMP-activated protein kinase (AMPK), a cellular metabolic sensor. Activated AMPK suppresses the expression of lipogenic enzymes and inhibits gluconeogenesis in the liver, reducing hepatic glucose output.
- Improvement of Insulin Sensitivity: It increases the translocation of GLUT4 transporters to the plasma membrane in peripheral muscle and adipose tissue, enhancing peripheral glucose uptake and utilization.
- Intestinal Glucose Absorption: Clinical data suggests a reduction in the intestinal absorption of glucose and an increase in GLP-1 (Glucagon-like peptide-1) secretion.
Pharmacokinetics (PK Profile)
Metformin exhibits unique pharmacokinetic properties compared to other oral antidiabetics, particularly regarding its lack of metabolism.
- Absorption: Slowly and incompletely absorbed from the gastrointestinal tract. Absolute bioavailability is approximately 50–60% under fasting conditions. Food decreases the extent of absorption and slightly delays the peak plasma concentration (Cmax).
- Distribution: Negligible plasma protein binding (<10%). Metformin partitions into erythrocytes, most likely as a function of time. The apparent volume of distribution (V/F) ranges from 654 ± 358 L due to significant tissue binding.
- Metabolism: No hepatic metabolism. Metformin is not metabolized by CYP450 isoenzymes and does not undergo conjugation. It is excreted unchanged in the urine.
- Excretion: Eliminated via renal tubular secretion and glomerular filtration. The elimination half-life (t1/2) is approximately 6.2 hours in plasma, but longer in the erythrocyte compartment. In patients with renal impairment, renal clearance decreases proportionally to creatinine clearance.
Clinical Indications (FDA Labeling)
Metformin is indicated as an adjunct to diet and exercise to improve glycemic control in adults and pediatric patients (10 years and older) with Type 2 Diabetes Mellitus.
Note: While widely studied for Polycystic Ovary Syndrome (PCOS) and gestational diabetes, these are considered off-label uses in many jurisdictions relative to the primary FDA approval.
Contraindications & Black Box Warnings
Regulatory bodies (FDA/EMA) emphasize specific safety protocols regarding renal function and metabolic states.
- Black Box Warning: Lactic Acidosis. Post-marketing cases of metformin-associated lactic acidosis have been reported, primarily in overdose scenarios or patients with severe renal impairment. This condition leads to hypothermia, hypotension, and resistant bradyarrhythmias.
- Renal Impairment: Contraindicated in patients with severe renal impairment (eGFR < 30 mL/min/1.73 m²). Initiation is not recommended if eGFR is 30–45 mL/min/1.73 m².
- Metabolic Acidosis: Contraindicated in acute or chronic metabolic acidosis, including diabetic ketoacidosis (DKA).
- Hypoxia & Sepsis: Conditions associated with hypoxemia or tissue hypoxia (e.g., acute heart failure, shock) increase the risk of lactic acidosis accumulation.