Clinical Definition
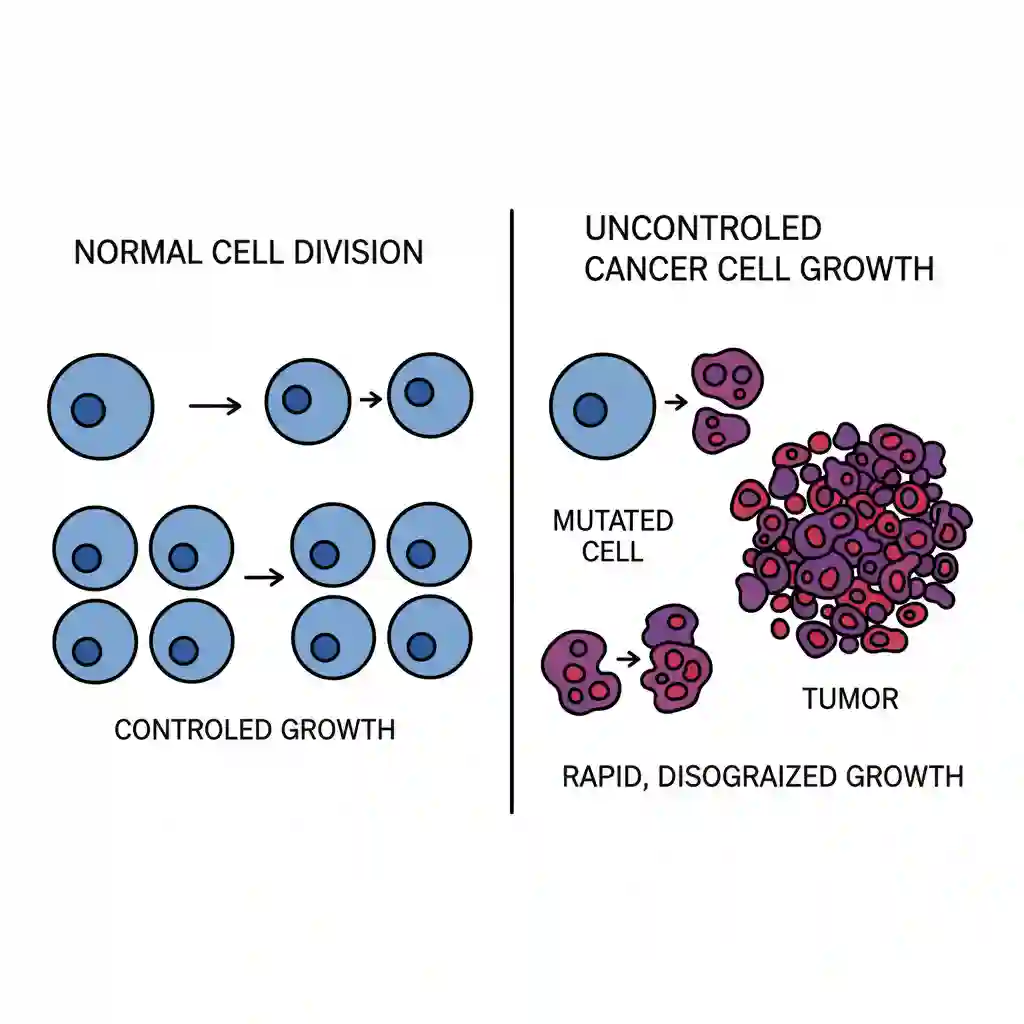
Malignant neoplasms (Cancer) constitute a broad and heterogeneous group of diseases characterized by uncontrolled cellular proliferation due to genomic instability. The defining features distinguishing malignancy from benign growths are the capacity for local tissue invasion and systemic dissemination (metastasis) via lymphatic or hematogenous routes. At a molecular level, the condition results from the accumulation of genetic and epigenetic alterations that disrupt the regulation of cell division and apoptosis (programmed cell death).
Clinical Coding & Classification
| System / Category | Code(s) | Description |
|---|---|---|
| ICD-10-CM | C00-C96 | Malignant neoplasms (Range) |
| ICD-10-CM | C80.1 | Malignant (primary) neoplasm, unspecified |
| ICD-O-3 | /3 | Morphology code suffix indicating Malignant (Primary Site) |
| CPT (Diagnostic) | 88305 | Tissue pathology, gross and microscopic examination |
| CPT (Therapeutic) | 96401-96549 | Chemotherapy and other highly complex drug or biologic agent administration |
Epidemiology & Statistics
Malignant neoplasms represent the second leading cause of death globally. According to Global Cancer Statistics (GLOBOCAN), the disease burden is increasing due to population aging and the adoption of cancer-associated lifestyle behaviors. The most incident types globally include breast, lung, colorectal, and prostate cancers. Mortality rates vary significantly by region, influenced by access to screening and therapeutic interventions.
Pathophysiology (Mechanism)
Carcinogenesis is a multi-step process driven by the acquisition of biological capabilities known as the “Hallmarks of Cancer.” Key mechanisms include:
1. Oncogene Activation: Gain-of-function mutations in proto-oncogenes (e.g., RAS, MYC) drive autonomous cell growth.
2. Tumor Suppressor Inactivation: Loss-of-function mutations in genes such as TP53 or RB1 remove cell cycle checkpoints and DNA repair mechanisms.
3. Angiogenesis: Tumors secrete factors like VEGF to stimulate the formation of new blood vessels, securing oxygen and nutrient supply.
4. Metastasis: Malignant cells undergo Epithelial-Mesenchymal Transition (EMT), allowing them to detach, invade the extracellular matrix, and colonize distant organs.
Standard Management Protocols
Treatment is multimodal, typically determined by the Tumour, Node, Metastasis (TNM) staging system and molecular profiling.
- Pharmacological Classes:
- Cytotoxic Chemotherapy: (e.g., Alkylating agents, Antimetabolites) These agents disrupt DNA synthesis or microtubule function, primarily targeting rapidly dividing cells.
- Targeted Therapy: (e.g., Tyrosine Kinase Inhibitors) Small molecules or antibodies designed to interfere with specific molecular targets involved in tumor growth (e.g., EGFR, HER2).
- Immune Checkpoint Inhibitors: (e.g., PD-1/PD-L1 inhibitors) Monoclonal antibodies that block immune-suppressive signaling, restoring the cytotoxic T-cell response against tumor cells.
- Surgical/Procedural Interventions:
- Resection: Curative intent surgery to remove the primary tumor mass and regional lymph nodes.
- Radiation Therapy: Use of high-energy ionizing radiation (e.g., IMRT, Proton Beam) to induce DNA damage in malignant tissue.
Healthcare Resource Utilization
Oncology care represents a substantial segment of healthcare expenditure. Resource utilization drivers include:
- Multidisciplinary Care: Management often requires coordination between surgical, medical, and radiation oncology departments (Tumor Boards).
- Advanced Imaging: Routine use of PET-CT and MRI for staging and surveillance.
- High-Cost Therapeutics: The introduction of biologics and CAR-T cell therapies has significantly increased the direct cost of pharmaceutical management.